What conditions must be met before a specific pathogen is proven to cause a disease?
Learning Objectives
- Summarize Koch'south postulates and molecular Koch'south postulates, respectively, and explain their significance and limitations
- Explicate the concept of pathogenicity (virulence) in terms of infectious and lethal dose
- Distinguish between principal and opportunistic pathogens and identify specific examples of each
- Summarize the stages of pathogenesis
- Explain the roles of portals of entry and exit in the manual of disease and identify specific examples of these portals
For virtually infectious diseases, the ability to accurately identify the causative pathogen is a critical step in finding or prescribing effective treatments. Today's physicians, patients, and researchers owe a sizable debt to the md Robert Koch (1843–1910), who devised a systematic arroyo for confirming causative relationships between diseases and specific pathogens.
Koch's Postulates
In 1884, Koch published iv postulates that summarized his method for determining whether a particular microorganism was the cause of a particular disease:
Koch'south Postulates
- The suspected pathogen must exist found in every instance of disease and not be found in good for you individuals.
- The suspected pathogen can be isolated and grown in pure culture.
- A good for you test subject infected with the suspected pathogen must develop the same signs and symptoms of affliction every bit seen in postulate i.
- The pathogen must be re-isolated from the new host and must be identical to the pathogen from postulate ii.
Each of Koch's postulates represents a benchmark that must be met before a illness can be positively linked with a pathogen. In guild to determine whether the criteria are met, tests are performed on laboratory animals and cultures from healthy and diseased animals are compared (Figure 1).
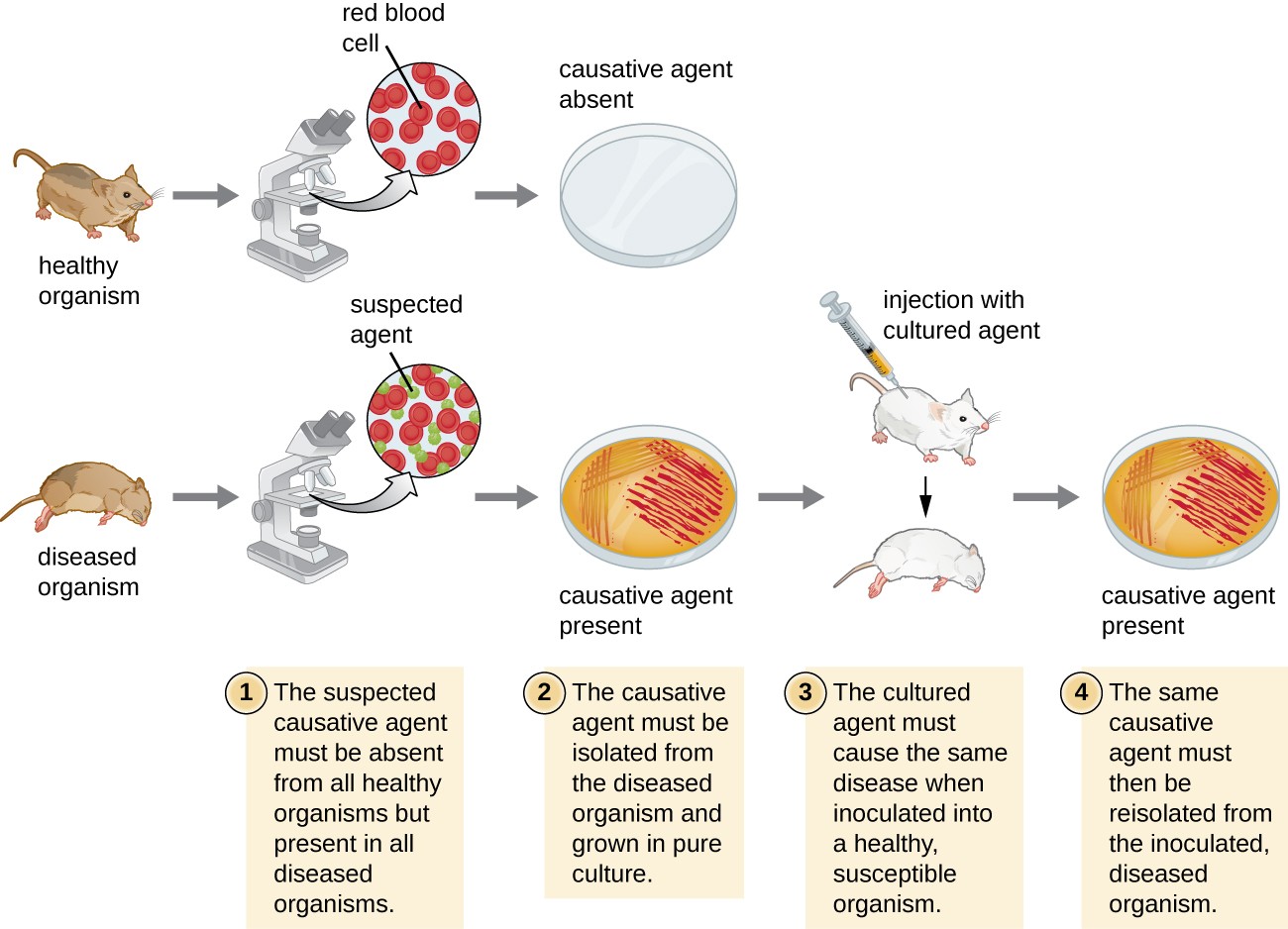
Figure 1. The steps for confirming that a pathogen is the cause of a particular illness using Koch's postulates.
In many ways, Koch'southward postulates are notwithstanding primal to our current understanding of the causes of disease. However, advances in microbiology have revealed some important limitations in Koch's criteria. Koch made several assumptions that we now know are untrue in many cases. The get-go relates to postulate 1, which assumes that pathogens are merely plant in diseased, not salubrious, individuals. This is non true for many pathogens. For example, H. pylori, described before in this chapter as a pathogen causing chronic gastritis, is also office of the normal microbiota of the breadbasket in many healthy humans who never develop gastritis. It is estimated that up of l% of the human population acquires H. pylori early on in life, with well-nigh maintaining it as function of the normal microbiota for the residuum of their life without ever developing affliction.
Koch's 2nd faulty assumption was that all healthy test subjects are equally susceptible to disease. We now know that individuals are not every bit susceptible to disease. Individuals are unique in terms of their microbiota and the state of their immune organization at any given time. The makeup of the resident microbiota can influence an private's susceptibility to an infection. Members of the normal microbiota play an important office in immunity by inhibiting the growth of transient pathogens. In some cases, the microbiota may prevent a pathogen from establishing an infection; in others, information technology may not forbid an infection birthday merely may influence the severity or blazon of signs and symptoms. As a event, ii individuals with the same disease may not ever nowadays with the same signs and symptoms. In addition, some individuals have stronger allowed systems than others. Individuals with immune systems weakened by age or an unrelated illness are much more susceptible to sure infections than individuals with strong immune systems.
Koch likewise assumed that all pathogens are microorganisms that can be grown in pure culture (postulate ii) and that animals could serve equally reliable models for human illness. All the same, we now know that not all pathogens can be grown in pure culture, and many homo diseases cannot be reliably replicated in beast hosts. Viruses and certain leaner, including Rickettsia and Chlamydia , are obligate intracellular pathogens that can grow only when inside a host cell. If a microbe cannot exist cultured, a researcher cannot move past postulate 2. Likewise, without a suitable nonhuman host, a researcher cannot evaluate postulate 2 without deliberately infecting humans, which presents obvious upstanding concerns. AIDS is an case of such a disease because the human being immunodeficiency virus (HIV) simply causes illness in humans.
Remember about It
- Briefly summarize the limitations of Koch's postulates.
Molecular Koch's Postulates
In 1988, Stanley Falkow (1934–) proposed a revised class of Koch'southward postulates known as molecular Koch's postulates. These are listed in the left column of Tabular array 1. The premise for molecular Koch's postulates is non in the power to isolate a detail pathogen just rather to identify a factor that may crusade the organism to be pathogenic.
Falkow'southward modifications to Koch's original postulates explain not only infections caused past intracellular pathogens merely likewise the existence of pathogenic strains of organisms that are usually nonpathogenic. For example, the predominant form of the bacterium Escherichia coli is a member of the normal microbiota of the human being intestine and is generally considered harmless. However, there are pathogenic strains of E. coli such as enterotoxigenic E. coli (ETEC) and enterohemorrhagic Due east. coli (O157:H7) (EHEC). We now know ETEC and EHEC exist because of the acquisition of new genes by the once-harmless East. coli, which, in the form of these pathogenic strains, is now capable of producing toxins and causing disease. The pathogenic forms resulted from modest genetic changes. The right-side cavalcade of Tabular array 1 illustrates how molecular Koch'due south postulates can be applied to identify EHEC as a pathogenic bacterium.
| Table ane. Molecular Koch'southward Postulates Applied to EHEC | |
|---|---|
| Molecular Koch's Postulates | Application to EHEC |
| (ane) The phenotype (sign or symptom of affliction) should be associated only with pathogenic strains of a species. | EHEC causes intestinal inflammation and diarrhea, whereas nonpathogenic strains of Eastward. coli do not. |
| (2) Inactivation of the suspected gene(s) associated with pathogenicity should result in a measurable loss of pathogenicity. | One of the genes in EHEC encodes for Shiga toxin, a bacterial toxin (toxicant) that inhibits poly peptide synthesis. Inactivating this gene reduces the bacteria's power to cause disease. |
| (3) Reversion of the inactive gene should restore the disease phenotype. | Past calculation the gene that encodes the toxin back into the genome (e.g., with a phage or plasmid), EHEC's ability to cause disease is restored. |
Every bit with Koch'due south original postulates, the molecular Koch'due south postulates accept limitations. For example, genetic manipulation of some pathogens is not possible using current methods of molecular genetics. In a similar vein, some diseases practice not take suitable animal models, which limits the utility of both the original and molecular postulates.
Recollect nearly It
- Explain the differences between Koch's original postulates and the molecular Koch'south postulates.
Pathogenicity and Virulence
The ability of a microbial agent to cause disease is called pathogenicity, and the degree to which an organism is pathogenic is called virulence. Virulence is a continuum. On one end of the spectrum are organisms that are avirulent (not harmful) and on the other are organisms that are highly virulent. Highly virulent pathogens will near always lead to a disease state when introduced to the torso, and some may even cause multi-organ and body system failure in healthy individuals. Less virulent pathogens may cause an initial infection, just may non e'er cause severe illness. Pathogens with low virulence would more than probable result in mild signs and symptoms of disease, such every bit depression-class fever, headache, or muscle aches. Some individuals might even be asymptomatic.
An example of a highly virulent microorganism is Bacillus anthracis , the pathogen responsible for anthrax. B. anthracis can produce different forms of disease, depending on the route of transmission (eastward.g., cutaneous injection, inhalation, ingestion). The almost serious course of anthrax is inhalation anthrax. Afterwards B. anthracis spores are inhaled, they germinate. An active infection develops and the bacteria release strong toxins that cause edema (fluid buildup in tissues), hypoxia (a condition preventing oxygen from reaching tissues), and necrosis (cell death and inflammation). Signs and symptoms of inhalation anthrax include high fever, difficulty breathing, airsickness and coughing upwards blood, and severe breast pains suggestive of a heart assail. With inhalation anthrax, the toxins and leaner enter the bloodstream, which tin can lead to multi-organ failure and decease of the patient. If a factor (or genes) involved in pathogenesis is inactivated, the bacteria become less virulent or nonpathogenic.
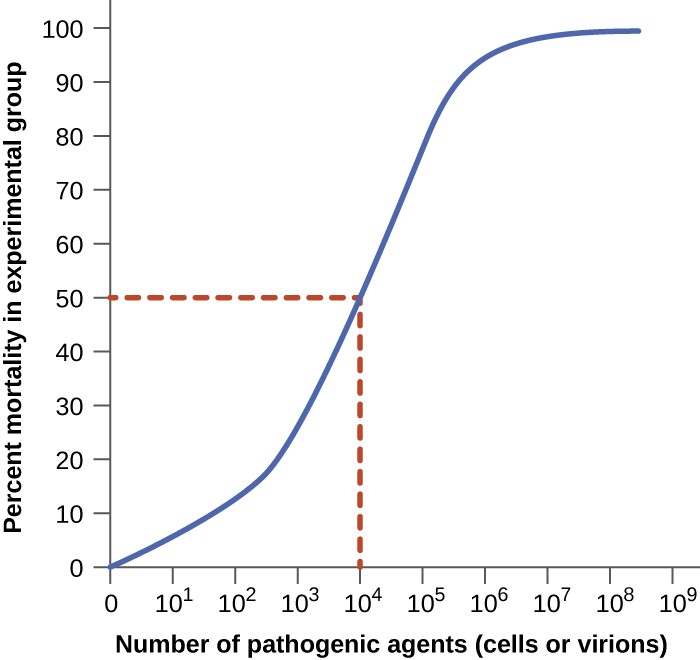
Figure 2. A graph similar this is used to make up one's mind LDfifty by plotting pathogen concentration against the percent of infected test animals that have died. In this example, the LD50 = ten4 pathogenic particles.
Virulence of a pathogen tin be quantified using controlled experiments with laboratory animals. Two important indicators of virulence are the median infectious dose (ID50) and the median lethal dose (LD50), both of which are typically determined experimentally using beast models. The ID50 is the number of pathogen cells or virions required to cause active infection in 50% of inoculated animals. The LD50 is the number of pathogenic cells, virions, or amount of toxin required to kill 50% of infected animals. To calculate these values, each group of animals is inoculated with one of a range of known numbers of pathogen cells or virions. In graphs like the i shown in Effigy ii, the percentage of animals that have been infected (for ID50) or killed (for LD50) is plotted against the concentration of pathogen inoculated. Figure two represents information graphed from a hypothetical experiment measuring the LDfifty of a pathogen. Interpretation of the data from this graph indicates that the LD50 of the pathogen for the examination animals is 10iv pathogen cells or virions (depending upon the pathogen studied).
Table 2 lists selected foodborne pathogens and their ID50 values in humans (every bit determined from epidemiologic data and studies on man volunteers). Keep in heed that these are median values. The actual infective dose for an individual can vary widely, depending on factors such as route of entry; the age, wellness, and immune status of the host; and environmental and pathogen-specific factors such as susceptibility to the acidic pH of the stomach. It is also important to note that a pathogen's infective dose does not necessarily correlate with affliction severity. For example, only a single jail cell of Salmonella enterica serotype Typhimurium tin can result in an active infection. The resultant disease, Salmonella gastroenteritis or salmonellosis, can cause nausea, airsickness, and diarrhea, simply has a mortality charge per unit of less than 1% in good for you adults. In contrast, S. enterica serotype Typhi has a much higher ID50, typically requiring every bit many as i,000 cells to produce infection. Nonetheless, this serotype causes typhoid fever, a much more systemic and severe affliction that has a mortality charge per unit as high equally 10% in untreated individuals.
| Table 2. ID50 for Selected Foodborne Diseases[one] | |
|---|---|
| Pathogen | ID50 |
| Viruses | |
| Hepatitis A virus | 10–100 |
| Norovirus | ane–ten |
| Rotavirus | ten–100 |
| Leaner | |
| Escherichia coli, enterohemorrhagic (EHEC, serotype O157) | x–100 |
| E. coli, enteroinvasive (EIEC) | 200–5,000 |
| E. coli, enteropathogenic (EPEC) | 10,000,000–x,000,000,000 |
| Eastward. coli, enterotoxigenic (ETEC) | 10,000,000–10,000,000,000 |
| Salmonella enterica serovar Typhi | <1,000 |
| S. enterica serovar Typhimurium | ≥1 |
| Shigella dysenteriae | 10–200 |
| Vibrio cholerae (serotypes O139, O1) | 1,000,000 |
| V. parahemolyticus | 100,000,000 |
| Protozoa | |
| Giardia lamblia | ane |
| Cryptosporidium parvum | 10–100 |
Recollect nigh It
- What is the difference between a pathogen's infective dose and lethal dose?
- Which is more closely related to the severity of a disease?
Master Pathogens versus Opportunistic Pathogens
Pathogens can be classified equally either primary pathogens or opportunistic pathogens. A primary pathogen tin can cause disease in a host regardless of the host's resident microbiota or immune system. An opportunistic pathogen, by contrast, can only crusade affliction in situations that compromise the host'due south defenses, such as the trunk's protective barriers, allowed arrangement, or normal microbiota. Individuals susceptible to opportunistic infections include the very young, the elderly, women who are meaning, patients undergoing chemotherapy, people with immunodeficiencies (such as acquired immunodeficiency syndrome [AIDS]), patients who are recovering from surgery, and those who have had a breach of protective barriers (such as a severe wound or burn).
An example of a primary pathogen is enterohemorrhagic E. coli (EHEC), which produces a virulence factor known as Shiga toxin. This toxin inhibits poly peptide synthesis, leading to severe and bloody diarrhea, inflammation, and renal failure, even in patients with healthy immune systems. Staphylococcus epidermidis , on the other mitt, is an opportunistic pathogen that is amongst the about frequent causes of nosocomial disease.[ii] Southward. epidermidis is a member of the normal microbiota of the peel, where information technology is more often than not avirulent. However, in hospitals, it can besides grow in biofilms that class on catheters, implants, or other devices that are inserted into the body during surgical procedures. In one case inside the body, Due south. epidermidis tin cause serious infections such as endocarditis, and it produces virulence factors that promote the persistence of such infections.
Other members of the normal microbiota tin can too cause opportunistic infections under certain weather. This oft occurs when microbes that reside harmlessly in one trunk location end up in a different body arrangement, where they cause disease. For example, Due east. coli normally found in the large intestine tin can cause a urinary tract infection if it enters the bladder. This is the leading cause of urinary tract infections among women.
Members of the normal microbiota may also crusade disease when a shift in the surround of the torso leads to overgrowth of a particular microorganism. For instance, the yeast Candida is part of the normal microbiota of the skin, oral fissure, intestine, and vagina, merely its population is kept in check by other organisms of the microbiota. If an individual is taking antibacterial medications, all the same, bacteria that would normally inhibit the growth of Candida can exist killed off, leading to a sudden growth in the population of Candida, which is non affected past antibacterial medications considering it is a mucus. An overgrowth of Candida can manifest as oral thrush (growth on mouth, throat, and tongue), a vaginal yeast infection, or cutaneous candidiasis. Other scenarios can also provide opportunities for Candida infections. Untreated diabetes can result in a high concentration of glucose in the saliva, which provides an optimal surround for the growth of Candida, resulting in thrush. Immunodeficiencies such as those seen in patients with HIV, AIDS, and cancer also lead to higher incidence of thrush. Vaginal yeast infections can result from decreases in estrogen levels during the menstruation or menopause. The amount of glycogen available to lactobacilli in the vagina is controlled past levels of estrogen; when estrogen levels are low, lactobacilli produce less lactic acid. The resultant increase in vaginal pH allows overgrowth of Candida in the vagina.
Call up about It
- Explain the difference between a primary pathogen and an opportunistic pathogen.
- Describe some weather condition nether which an opportunistic infection tin occur.
Stages of Pathogenesis
To cause disease, a pathogen must successfully achieve iv steps or stages of pathogenesis: exposure (contact), adhesion (colonization), invasion, and infection. The pathogen must exist able to proceeds entry to the host, travel to the location where it can plant an infection, evade or overcome the host'due south immune response, and cause damage (i.east., disease) to the host. In many cases, the cycle is completed when the pathogen exits the host and is transmitted to a new host.
Exposure
An encounter with a potential pathogen is known as exposure or contact. The food we eat and the objects we handle are all means that we tin can come up into contact with potential pathogens. Yet, not all contacts effect in infection and disease. For a pathogen to cause affliction, it needs to be able to gain access into host tissue. An anatomic site through which pathogens can pass into host tissue is called a portal of entry. These are locations where the host cells are in direct contact with the external surroundings. Major portals of entry are identified in Effigy 3 and include the skin, mucous membranes, and parenteral routes.

Figure 3. Shown are different portals of entry where pathogens can proceeds access into the body. With the exception of the placenta, many of these locations are direct exposed to the external environment.
Mucosal surfaces are the well-nigh important portals of entry for microbes; these include the mucous membranes of the respiratory tract, the alimentary canal, and the genitourinary tract. Although most mucosal surfaces are in the interior of the trunk, some are contiguous with the external skin at various trunk openings, including the eyes, olfactory organ, rima oris, urethra, and anus.
Most pathogens are suited to a particular portal of entry. A pathogen's portal specificity is determined by the organism'south environmental adaptions and past the enzymes and toxins they secrete. The respiratory and gastrointestinal tracts are particularly vulnerable portals of entry because particles that include microorganisms are constantly inhaled or ingested, respectively.
Pathogens tin can too enter through a alienation in the protective barriers of the skin and mucous membranes. Pathogens that enter the body in this way are said to enter by the parenteral route. For example, the pare is a proficient natural barrier to pathogens, but breaks in the skin (e.k., wounds, insect bites, animal bites, needle pricks) can provide a parenteral portal of entry for microorganisms.
In pregnant women, the placenta normally prevents microorganisms from passing from the mother to the fetus. All the same, a few pathogens are capable of crossing the blood-placental bulwark. The gram-positive bacterium Listeria monocytogenes , which causes the foodborne disease listeriosis, is one instance that poses a serious risk to the fetus and tin sometimes lead to spontaneous abortion. Other pathogens that tin pass the placental barrier to infect the fetus are known collectively by the acronym TORCH (Tabular array 3).
Transmission of infectious diseases from female parent to baby is also a concern at the time of birth when the babe passes through the birth canal. Babies whose mothers take active chlamydia or gonorrhea infections may be exposed to the causative pathogens in the vagina, which can result in eye infections that lead to blindness. To foreclose this, it is standard practice to administer antibody drops to infants' eyes shortly after nascency.
| Table 3. Pathogens Capable of Crossing the Placental Barrier (TORCH Infections) | ||
|---|---|---|
| Disease | Pathogen | |
| T | Toxoplasmosis | Toxoplasma gondii (protozoan) |
| O [3] | Syphilis Chickenpox Hepatitis B HIV Fifth disease (erythema infectiosum) | Treponema pallidum (bacterium) Varicella-zoster virus (human herpesvirus iii) Hepatitis B virus (hepadnavirus) Retrovirus Parvovirus B19 |
| R | Rubella (German measles) | Togavirus |
| C | Cytomegalovirus | Human herpesvirus 5 |
| H | Canker | Canker simplex viruses (HSV) one and 2 |
Clinical Focus: Pankaj, Part 2
This example continues Pankaj's story that started in Characteristics of Communicable diseases.
At the clinic, a physician takes down Pankaj's medical history and asks nigh his activities and diet over the past calendar week. Upon learning that Pankaj became ill the 24-hour interval subsequently the party, the md orders a blood exam to check for pathogens associated with foodborne diseases. After tests confirm that presence of a gram-positive rod in Pankaj'southward claret, he is given an injection of a broad-spectrum antibiotic and sent to a nearby hospital, where he is admitted equally a patient. There he is to receive additional intravenous antibiotic therapy and fluids.
- Is this bacterium in Pankaj's claret function of normal microbiota?
- What portal of entry did the bacteria use to cause this infection?
Nosotros'll return to Pankaj'southward example in later on pages.
Adhesion
Post-obit the initial exposure, the pathogen adheres at the portal of entry. The term adhesion refers to the capability of pathogenic microbes to attach to the cells of the body using adhesion factors, and different pathogens use various mechanisms to attach to the cells of host tissues.

Figure 4. Glycocalyx produced past bacteria in a biofilm allows the cells to attach to host tissues and to medical devices such as the catheter surface shown here. (credit: modification of work past Centers for Affliction Control and Prevention)
Molecules (either proteins or carbohydrates) called adhesins are found on the surface of certain pathogens and bind to specific receptors (glycoproteins) on host cells. Adhesins are present on the fimbriae and flagella of bacteria, the cilia of protozoa, and the capsids or membranes of viruses. Protozoans can likewise apply hooks and barbs for adhesion; spike proteins on viruses also enhance viral adhesion. The production of glycocalyces (slime layers and capsules) (Effigy 4), with their high carbohydrate and poly peptide content, can also allow certain bacterial pathogens to attach to cells.
Biofilm growth can also act as an adhesion gene. A biofilm is a community of leaner that produce a glycocalyx, known every bit extrapolymeric substance (EPS), that allows the biofilm to attach to a surface. Persistent Pseudomonas aeruginosa infections are common in patients suffering from cystic fibrosis, fire wounds, and middle-ear infections (otitis media) considering P. aeruginosa produces a biofilm. The EPS allows the bacteria to adhere to the host cells and makes information technology harder for the host to physically remove the pathogen. The EPS not simply allows for attachment just provides protection confronting the immune system and antibody treatments, preventing antibiotics from reaching the bacterial cells within the biofilm. In addition, not all leaner in a biofilm are rapidly growing; some are in stationary phase. Since antibiotics are near constructive against rapidly growing bacteria, portions of bacteria in a biofilm are protected confronting antibiotics.[four]
Invasion
Once adhesion is successful, invasion can continue. Invasion involves the dissemination of a pathogen throughout local tissues or the body. Pathogens may produce exoenzymes or toxins, which serve as virulence factors that permit them to colonize and damage host tissues as they spread deeper into the body. Pathogens may also produce virulence factors that protect them against immune organization defenses. A pathogen's specific virulence factors determine the degree of tissue damage that occurs. Figure v shows the invasion of H. pylori into the tissues of the stomach, causing damage every bit it progresses.

Figure 5. H. pylori is able to invade the lining of the tum by producing virulence factors that enable it pass through the mucin layer covering epithelial cells. (credit: modification of work by Zina Deretsky, National Science Foundation)
Intracellular pathogens achieve invasion by entering the host'southward cells and reproducing. Some are obligate intracellular pathogens (meaning they can only reproduce inside of host cells) and others are facultative intracellular pathogens (meaning they can reproduce either inside or outside of host cells). Past entering the host cells, intracellular pathogens are able to evade some mechanisms of the allowed organisation while also exploiting the nutrients in the host cell.
Entry to a cell tin can occur by endocytosis. For nearly kinds of host cells, pathogens apply one of 2 unlike mechanisms for endocytosis and entry. One machinery relies on effector proteins secreted past the pathogen; these effector proteins trigger entry into the host cell. This is the method that Salmonella and Shigella use when invading intestinal epithelial cells. When these pathogens come in contact with epithelial cells in the intestine, they secrete effector molecules that cause protrusions of membrane ruffles that bring the bacterial cell in. This process is called membrane ruffling. The 2d mechanism relies on surface proteins expressed on the pathogen that bind to receptors on the host jail cell, resulting in entry. For example, Yersinia pseudotuberculosis produces a surface protein known as invasin that binds to beta-i integrins expressed on the surface of host cells.
Some host cells, such every bit white blood cells and other phagocytes of the immune system, actively endocytose pathogens in a process called phagocytosis. Although phagocytosis allows the pathogen to gain entry to the host jail cell, in most cases, the host cell kills and degrades the pathogen by using digestive enzymes. Commonly, when a pathogen is ingested by a phagocyte, it is enclosed within a phagosome in the cytoplasm; the phagosome fuses with a lysosome to grade a phagolysosome, where digestive enzymes impale the pathogen (see Pathogen Recognition and Phagocytosis). Withal, some intracellular pathogens have the ability to survive and multiply within phagocytes. Examples include Listeria monocytogenes and Shigella ; these bacteria produce proteins that lyse the phagosome before information technology fuses with the lysosome, allowing the bacteria to escape into the phagocyte's cytoplasm where they can multiply. Leaner such equally Mycobacterium tuberculosis , Legionella pneumophila , and Salmonella species use a slightly different mechanism to evade being digested past the phagocyte. These bacteria prevent the fusion of the phagosome with the lysosome, thus remaining live and dividing within the phagosome.
Infection
Following invasion, successful multiplication of the pathogen leads to infection. Infections tin can be described as local, focal, or systemic, depending on the extent of the infection. A local infection is confined to a small surface area of the body, typically well-nigh the portal of entry. For example, a hair follicle infected by Staphylococcus aureus infection may result in a eddy effectually the site of infection, but the bacterium is largely contained to this small location. Other examples of local infections that involve more all-encompassing tissue interest include urinary tract infections confined to the float or pneumonia bars to the lungs.
In a focal infection, a localized pathogen, or the toxins information technology produces, can spread to a secondary location. For case, a dental hygienist nicking the gum with a sharp tool can lead to a local infection in the mucilage by Streptococcus leaner of the normal oral microbiota. These Streptococcus spp. may and so gain access to the bloodstream and brand their way to other locations in the body, resulting in a secondary infection.
When an infection becomes disseminated throughout the body, we call it a systemic infection. For case, infection past the varicella-zoster virus typically gains entry through a mucous membrane of the upper respiratory organization. It and so spreads throughout the body, resulting in the archetype cerise pare lesions associated with chickenpox. Since these lesions are non sites of initial infection, they are signs of a systemic infection.
Sometimes a main infection, the initial infection caused by ane pathogen, can lead to a secondary infection past another pathogen. For case, the immune system of a patient with a primary infection by HIV becomes compromised, making the patient more susceptible to secondary diseases like oral thrush and others caused by opportunistic pathogens. Similarly, a chief infection by Influenzavirus damages and decreases the defense mechanisms of the lungs, making patients more susceptible to a secondary pneumonia by a bacterial pathogen similar Haemophilus influenzae or Streptococcus pneumoniae . Some secondary infections can even develop equally a result of treatment for a primary infection. Antibiotic therapy targeting the main pathogen can cause collateral damage to the normal microbiota, creating an opening for opportunistic pathogens.
A Secondary Yeast Infection
Anita, a 36-year-erstwhile mother of 3, goes to an urgent care center complaining of pelvic pressure, frequent and painful urination, abdominal cramps, and occasional claret-tinged urine. Suspecting a urinary tract infection (UTI), the physician requests a urine sample and sends it to the lab for a urinalysis. Since it will have approximately 24 hours to get the results of the culturing, the physician immediately starts Anita on the antibody ciprofloxacin. The next 24-hour interval, the microbiology lab confirms the presence of E. coli in Anita'southward urine, which is consistent with the presumptive diagnosis. Notwithstanding, the antimicrobial susceptibility test indicates that ciprofloxacin would not effectively care for Anita's UTI, so the doctor prescribes a different antibiotic.
After taking her antibiotics for ane calendar week, Anita returns to the dispensary complaining that the prescription is non working. Although the painful urination has subsided, she is now experiencing vaginal itching, burning, and discharge. After a brief test, the physician explains to Anita that the antibiotics were likely successful in killing the Due east. coli responsible for her UTI; however, in the process, they also wiped out many of the "skilful" bacteria in Anita'due south normal microbiota. The new symptoms that Anita has reported are consequent with a secondary yeast infection past Candida albicans, an opportunistic mucus that normally resides in the vagina but is inhibited past the bacteria that usually reside in the same surround.
To ostend this diagnosis, a microscope slide of a directly vaginal smear is prepared from the discharge to check for the presence of yeast. A sample of the discharge accompanies this slide to the microbiology lab to determine if there has been an increment in the population of yeast causing vaginitis. After the microbiology lab confirms the diagnosis, the doctor prescribes an antifungal drug for Anita to employ to eliminate her secondary yeast infection.
- Why was Candida not killed by the antibiotics prescribed for the UTI?
Retrieve about It
- List three conditions that could pb to a secondary infection.
Transmission of Disease
For a pathogen to persist, it must put itself in a position to be transmitted to a new host, leaving the infected host through a portal of leave (Figure 6). Every bit with portals of entry, many pathogens are adapted to use a item portal of leave. Similar to portals of entry, the most common portals of exit include the pare and the respiratory, urogenital, and gastrointestinal tracts. Coughing and sneezing tin can expel pathogens from the respiratory tract. A single sneeze can send thousands of virus particles into the air. Secretions and excretions can transport pathogens out of other portals of get out. Feces, urine, semen, vaginal secretions, tears, sweat, and shed peel cells tin can all serve as vehicles for a pathogen to leave the trunk. Pathogens that rely on insect vectors for transmission get out the trunk in the blood extracted by a biting insect. Similarly, some pathogens exit the body in blood extracted past needles.

Figure 6. Pathogens leave the torso of an infected host through various portals of leave to infect new hosts.
Key Concepts and Summary
- Koch's postulates are used to make up one's mind whether a particular microorganism is a pathogen. Molecular Koch's postulates are used to determine what genes contribute to a pathogen'south power to cause disease.
- Virulence, the degree to which a pathogen can cause affliction, can be quantified past calculating either the ID50 or LDl of a pathogen on a given population.
- Primary pathogens are capable of causing pathological changes associated with disease in a healthy individual, whereas opportunistic pathogens tin but cause disease when the private is compromised past a break in protective barriers or immunosuppression.
- Infections and illness can be caused by pathogens in the environment or microbes in an individual's resident microbiota.
- Infections can exist classified as local, focal, or systemic depending on the extent to which the pathogen spreads in the body.
- A secondary infection can sometimes occur afterward the host's defenses or normal microbiota are compromised by a primary infection or antibody treatment.
- Pathogens enter the torso through portals of entry and leave through portals of go out. The stages of pathogenesis include exposure, adhesion, invasion, infection, and transmission.
Multiple Pick
Which of the following is a pathogen that could not be identified by the original Koch'due south postulates?
- Staphylococcus aureus
- Pseudomonas aeruginosa
- Man immunodeficiency virus
- Salmonella enterica serovar Typhimurium
Show Answer
Reply c. Homo immunodeficiency virus could not be identified past the original Koch's postulates.
Pathogen A has an ID50 of 50 particles, pathogen B has an ID50 of 1,000 particles, and pathogen C has an ID50 of 1 × 106 particles. Which pathogen is virtually virulent?
- pathogen A
- pathogen B
- pathogen C
Show Answer
Reply a. Pathogen A is most virulent.
Which of the post-obit choices lists the steps of pathogenesis in the correct gild?
- invasion, infection, adhesion, exposure
- adhesion, exposure, infection, invasion
- exposure, adhesion, invasion, infection
- disease, infection, exposure, invasion
Show Answer
Reply c. The correct order is exposure, adhesion, invasion, and then infection.
Fill up in the Bare
A(n) __________ pathogen causes disease only when weather are favorable for the microorganism considering of transfer to an inappropriate body site or weakened amnesty in an individual.
Evidence Answer
Anopportunistic pathogen causes disease but when conditions are favorable for the microorganism because of transfer to an inappropriate trunk site or weakened immunity in an individual.
The concentration of pathogen needed to kill 50% of an infected group of test animals is the __________.
Show Respond
The concentration of pathogen needed to impale 50% of an infected grouping of test animals is the LDl .
A(n) __________ infection is a small region of infection from which a pathogen may movement to another part of the trunk to establish a second infection.
Bear witness Reply
A focal infection is a minor region of infection from which a pathogen may move to another part of the body to establish a second infection.
Cilia, fimbriae, and pili are all examples of structures used by microbes for __________.
Prove Answer
Cilia, fimbriae, and pili are all examples of structures used by microbes for adhesion.
Think well-nigh It
- Diseases that involve biofilm-producing bacteria are of serious concern. They are not equally hands treated compared with those involving costless-floating (or planktonic) bacteria. Explain three reasons why biofilm formers are more pathogenic.
- A microbiologist has identified a new gram-negative pathogen that causes liver disease in rats. She suspects that the bacterium'south fimbriae are a virulence factor. Describe how molecular Koch's postulates could be used to test this hypothesis.
- Acupuncture is a form of alternative medicine that is used for pain relief. Explain how acupuncture could facilitate exposure to pathogens.

Source: https://courses.lumenlearning.com/microbiology/chapter/how-pathogens-cause-disease/
0 Response to "What conditions must be met before a specific pathogen is proven to cause a disease?"
Post a Comment